Background
Hypertension has high global prevalence with suboptimal control rates. In Lebanon, its prevalence is estimated to around 37%. Antihypertensive monotherapy proved to have limited effectiveness in reducing blood pressure. A commonly used combination of irbesartan, an angiotensin ii receptor blocker, and amlodipine, a calcium channel blocker, has highly potent antihypertensive effects.
Objective
The aim of this study was to assess the safety profile of irbesartan/amlodipine fixed-dose combination (FDC) in real-life practice, and to evaluate its use and its effectiveness in reducing blood pressure (BP) in the Lebanese population.
Patients and methods
A total of 603 patients, who provided written consent, were enrolled in this national, multicenter, non-interventional, longitudinal product registry, and data was collected at baseline and then at 3 follow-up visits separated by a period of 3 months.
Results
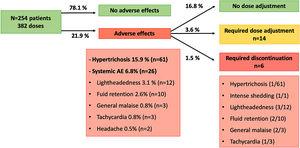
More than 80% of the patients were overweight or obese and 65% of them presented comorbidities, mainly dyslipidemia. Adverse events (AEs) were reported in around 12% of the cases, most of which were non-serious. Treatment-emergent (TE) AEs were experienced by around 6% of the patients, and none of the serious AEs were TE. BP level was significantly reduced at 3 months and 9 months after baseline. Around 80% of patients achieved their target BP at 3 months post-baseline. Patients’ weight and heart rate also sequentially decreased throughout the visits.
Conclusion
This study provides a better understanding of patients with hypertension treated with irbesartan-amlodipine FDC in Lebanon, and confirms the good safety and tolerability profile of this anti-hypertensive FDC.