Introduction
The anti-CD20 monoclonal antibodies, rituximab (RTX) and ocrelizumab (OCRE), are used routinely for the treatment of neuroimmunological diseases. However, infusion-related reactions (IRRs) are common, and their underlying mechanisms are not well defined.
Objectives
Our aim was to determine the incidence of IRRs during OCRE and RTX treatment for neuroimmunological diseases and to identify predisposing factors.
Methods
We conducted a retrospective observational study including patients who received at least one dose of OCRE or RTX between October 2018 and March 2022. Demographic data, comorbidities, immunological variables, and information about treatments and IRRs were collected. Statistical analysis was performed to assess IRR incidence, differences between groups, and predictors of IRR.
Results
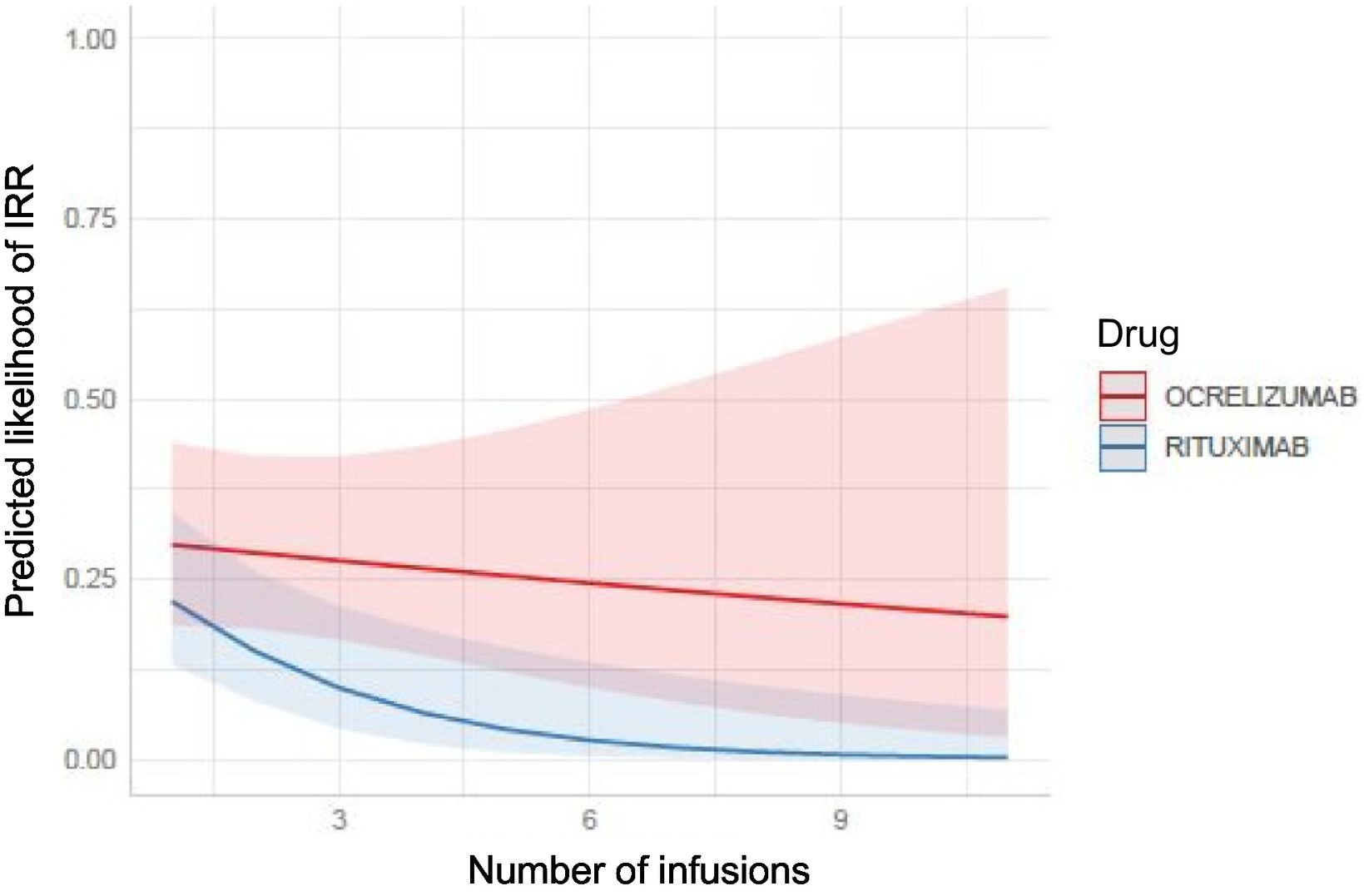
Of 48 patients receiving 239 infusions, 17.6% presented IRRs. The incidence of IRRs was 40% among all patients: 41% in the OCRE group and 38% in the RTX group. Most IRRs were mild (90%). We found statistically significant differences in infusion time between both groups, with longer times associated with higher IRR incidence. Interestingly, a higher percentage of B lymphocytes prior to infusion was associated with increased risk of IRRs.
Conclusion
IRRs are common with both treatments, especially in the early stages. The percentage of B lymphocytes before infusion and longer infusion times are associated with a higher risk of developing IRRs. These findings may help to mitigate IRRs during treatment.